/GettyImages-170075160-56a133b73df78cf772685a14.jpg)
These subatomic particles are composed of even smaller particles known as quarks which came together after the Big Bang around 13.7 million years ago. The nucleus is surrounded by a cloud of electrons at varying energy levels. In 1913, Bohr presented a revolutionary quantum atom based on. Atomic Structure: All atoms consist of a nucleus which contains two types of subatomic particles - protons and neutrons.
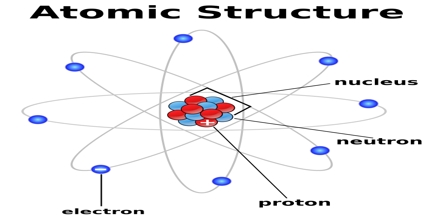
The heaviest stable atom is lead-208, with a mass of 207.976 6521 Da. However, protons have a charge of +1, and neutrons are uncharged. In the early 20th century, many models of the internal atomic structure were proposed. A given atom has an atomic mass approximately equal (within 1) to its mass number times the atomic mass unit (for example the mass of a nitrogen-14 is roughly 14 Da), but this number will not be exactly an integer except (by definition) in the case of carbon-12. Protons and neutrons are collectively known as nucleons. \( \newcommand\): Protons, neutrons, and electrons: Both protons and neutrons have a mass of 1 amu and are found in the nucleus. The nucleus is at the centre of the atom and contains the protons and neutrons.
